Mobile CT scanner SOMATOM On.site from Siemens gets FDA Clearance
Now head imaging of critically ill patients by enabling patients and staff to remain in ICU
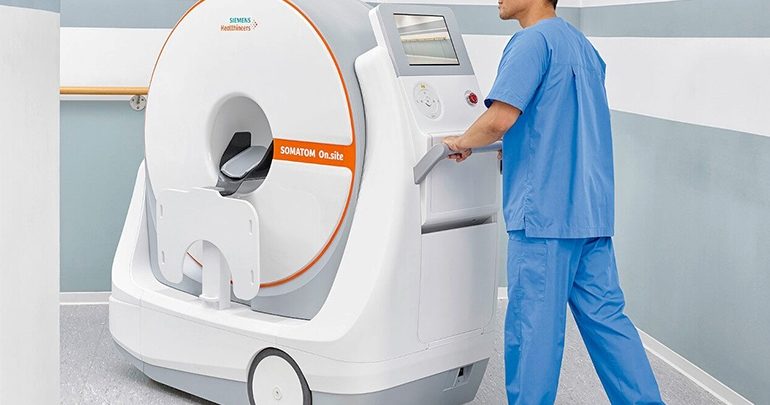
Malvern, Pennsylvania: The US Food and Drug Administration (FDA) has cleared a mobile head computed tomography (CT) scanner from Siemens Healthineers that enables a critically ill patient to receive CT head imaging in the intensive care unit (ICU) while remaining in bed.
Douglas Ryan, Vice President, CT, Siemens Healthineers North America said, “The system delivers reliable and consistent image quality demanded by healthcare professionals in the ICU, neurology, and radiology departments. Additionally, bedside imaging helps to reduce patient transports, thereby reducing the risk of infection while improving workforce efficiency.”
Called the SOMATOM On.site, the CT enables reliable and consistent image quality at the point of care, allowing customers to transform care delivery. The SOMATOM On.site system design – with the telescopic, self-shielded gantry and attachable front and back radiation shields – reduces scatter radiation compared to CT scanners without this design and provides radiation protection for neighboring patients and staff.




