Sun Pharma announces publication of ILUMYA’s five-year efficacy results
The results were announced at British Journal of Dermatology
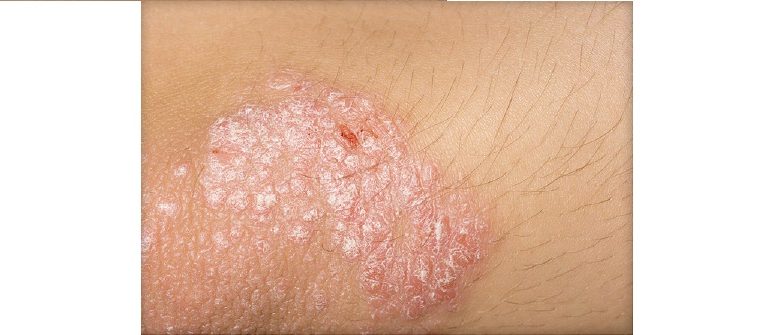
Sun Pharmaceutical Industries, USA (Sun Pharma) announced the publication of five-year Phase 3 efficacy and safety results for ILUMYA (tildrakizumab-asmn) based on a pooled analysis of the ReSURFACE 1 and ReSURFACE 2 extension studies in the British Journal of Dermatology. Patients with moderate-to-severe plaque psoriasis who received ILUMYA maintained consistent, high levels of skin clearance with no new safety signals reported through five years of continuous treatment. This is the longest complete dataset of an IL-23 inhibitor reported to date with a total ILUMYA exposure of more than 5,400 patient-years.
In an analysis of the pooled reSURFACE 1 and reSURFACE 2 extension study data, patients could receive ILUMYA 100 mg or 200 mg through five years of continuous treatment. ILUMYA 100 mg is approved in the US, Japan and Australia, and 200 mg is additionally approved under the brand name ILUMETRI in Europe. The results showed moderate-to-severe plaque psoriasis patients who responded at Week 28 had well-maintained efficacy as assessed by relative clinical improvement and extent of disease activity through Week 244. In patients who have treated with ILUMYA 100 mg, the median improvement from baseline Psoriasis Area and Severity Index (PASI) score was 95.5 per cent and 65.5 per cent of patients achieved Physician’s Global Assessment (PGA) 0/1 at Week 244. Furthermore, patients achieved a median absolute PASI score of 1.1 on the 72-point PASI disease activity scale at Week 244, which indicates patients treated with ILUMYA had low residual disease following treatment. Absolute PASI scores are used in clinical trials to access efficacy and can provide an indication of the extent of residual disease after treatment.
ILUMYA 100 mg was well-tolerated during the Phase 3 trials. The three adverse reactions (AEs) that occurred more frequently than placebo and ≥1% in clinical trials were upper respiratory infections (14 per cent vs. 12 per cent), injection site reactions (3 per cent vs. 2 per cent) and diarrhoea (2 per cent vs. 1 per cent). Furthermore, the incidence rates of severe infections, malignancies, major adverse cardiovascular events, and AEs in patients over 65 years of age, were comparable throughout five years of treatment, and no new safety signals were observed.
“We are proud to publish five-year data reinforcing our understanding that a high level of sustained skin clearance and a durable safety profile is achievable with ILUMYA, regardless of baseline level of skin disease, age or background illnesses,” said Abhay Gandhi, CEO, Sun Pharma, North America. “These impressive results underscore our commitment to patients living with moderate-to-severe plaque psoriasis by providing further assurance of the long-term efficacy and safety of ILUMYA.”
ILUMYA is approved for adults with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy and is being evaluated for other possible uses.




