Drug Controller General of India (DCGI)
-
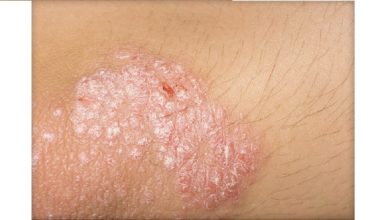
Eli Lilly launches psoriasis drug Copellor (Ixekizumab) in India
Copellor (Ixekizumab) is a prescription medicine to be used only on the advice of a dermatologist or rheumatologist and under…
Read More » -
Alkem collaborates with Stempeutics for knee osteoarthritis product StemOne
The treatment comprises a single intra-articular injection of StemOne in an outpatient procedure with no anaesthesia requirement Alkem Laboratories announced…
Read More » -
Molbio launches Truenat Scrub T test for Scrub Typhus
A chip-based, multi-disease platform can test over 30 diseases Molbio Diagnostics, the innovator of Truenat, a point of care Real-Time…
Read More » -
Enzene Biosciences obtains MA for Romiplostim Biosimilar drug in India
The drug is indicated for the treatment of chronic Immune Thrombocytopenic Purpura (ITP) in adults Enzene Biosciences ‘Enzene’ announced a…
Read More » -
PNB Vesper completes COVID-19 Phase 2 Clinical Trial
Submits report to DCGI and Government of India for Emergency Use Authorisation Kerala-based life sciences company PNB Vesper announced that…
Read More » -
Bharat Biotech to start Phase-1 trials of COVID-19 intranasal vaccine in Feb-March
Phase I human clinical trials will be conducted in India Besides Covaxin, Bharat Biotech has been actively working on developing…
Read More »