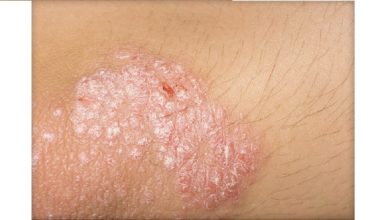
psoriasis
-
Lord’s Mark Biotech launches Tinefcon for psoriasis management
Has set a revenue target of Rs 100 crores for Tinefcon by 2025 Lord’s Mark Biotech, a subsidiary of Lord’s…
Read More » -
SPEVIGO bags breakthrough therapy designation in US for prevention of generalised pustular psoriasis flares
The designation follows the Center for Drug Evaluation (CDE) of the China National Medical Products Administration (NMPA), which also recently…
Read More » -
Eli Lilly launches psoriasis drug Copellor (Ixekizumab) in India
Copellor (Ixekizumab) is a prescription medicine to be used only on the advice of a dermatologist or rheumatologist and under…
Read More » -
Bristol Myers psoriasis drug succeeds in late-stage study
The drug, deucravacitinib, met both main goals of the study by reducing the extent and severity of psoriasis after 16…
Read More » -
Sun Pharma announces sustained efficacy for ILUMYA
ILUMYA is the first IL-23 inhibitor to complete five years of study based on a pooled analysis of two Phase…
Read More »