FDA Clears the Eko DUO Cardiac Monitoring Device
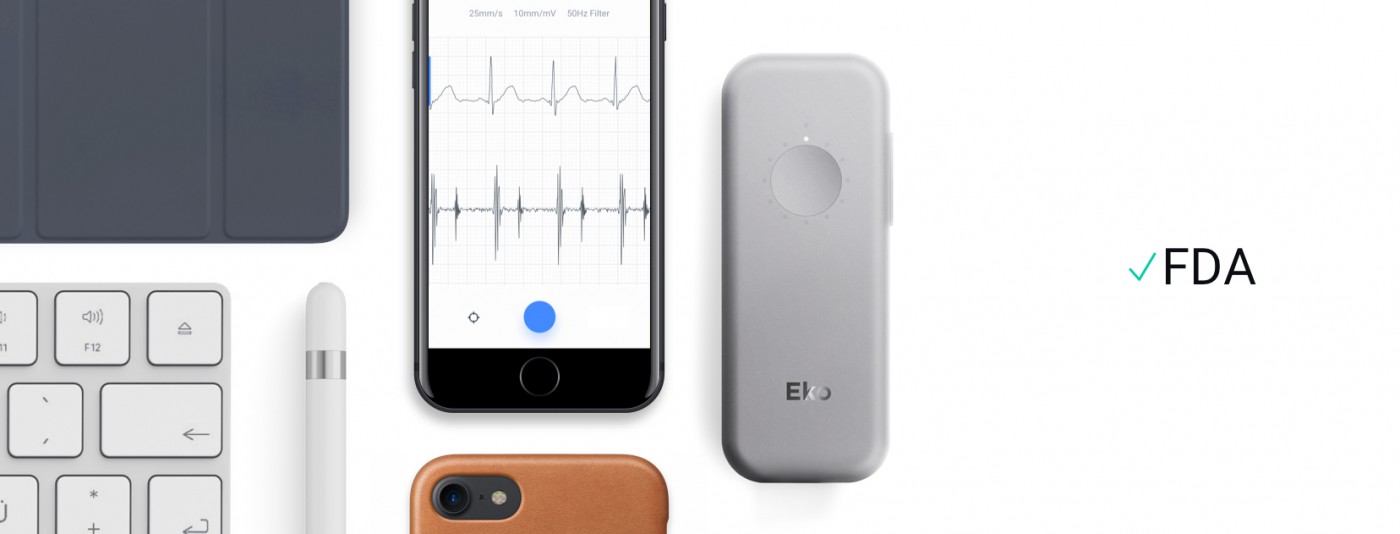
Eko Devices, the leader in mobile acoustic cardiac monitoring tools, has received FDA clearance to market its latest innovation, DUO – a combined digital stethoscope and electrocardiogram (ECG). The portable cardiac device was inspired by cardiologists’ demand for more effective monitoring tools for heart disease the press release said.
The marriage of ECG and digital stethoscope technology into a compact, handheld device offers unprecedented insight into cardiac function. Clinicians will use DUO as a cutting-edge screening tool – or prescribe it to heart disease outpatients as the centerpiece of at-home health kits. The device wirelessly pairs with Eko’s secure, HIPAA-compliant app, enabling remote monitoring and diagnosis by a clinician or specialist.
“Eko DUO’s consumer-friendly design can help transform how clinicians monitor heart health in-person or virtually,” said Dr. John Chorba, a Cardiologist and Assistant Professor at UC San Francisco. “We need powerful tools which heart failure patients can use to improve self-care and communicate troubling findings with an expert.”
DUO represents a shift in the fight against heart disease, empowering patients to use familiar smart tools under the remote supervision of their physician to navigate complex medical conditions and, ultimately, reduce unnecessary hospital re-admissions. Studies reveal that 25 percent of heart failure patients are re-admitted to the hospital within 30 days and 50 percent are re-admitted within six months. Heart failure costs the nation $30.7 billion annually.
“Cardiology programs are looking for ways to deliver hospital-quality healthcare at home” said Dr. Ami Bhatt, Director of Outpatient Cardiology and the Adult Congenital Heart Disease Program at Massachusetts General Hospital. “The ability to capture digital heart sounds and an ECG expands our portfolio of mechanisms to remotely monitor the heart – and brings diagnosis and opportunities for early intervention even further upstream. Robust toolkits for caring for patients in the community will hopefully lead to more appropriate healthcare utilization through continuous rather than episodic outpatient care.”
Following its vision to improve cardiovascular care, Eko is also developing machine-learning algorithms that can be combined with DUO to automatically alert patients and their care teams of suspected decline in cardiac function.
“Our mission is to make cardiac monitoring more accessible for patients and physicians to ultimately reduce unnecessary readmissions and reduce the excessive healthcare burden of heart disease,” said Connor Landgraf, co-founder and CEO of Eko. “The launch of DUO and our upcoming decision-support algorithms are giant steps forward in patient empowerment and telemedicine. It’s important to get this in the hands of the millions of heart disease patients who deserve better at-home monitoring tools.”




